Clinical tests for malaria vaccine now in phase III
Summary
RTS,S is the first vaccine to provide protection against parasitic diseases.
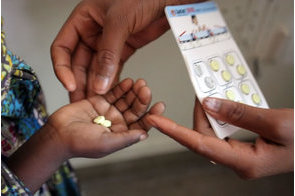
Clinical trials of RTS,S, an anti-malaria vaccine, are ongoing in seven African countries with quite promising results. Initial results have indicated that the vaccine reduces a child’s likelihood of falling ill with malaria by half. RTS,S is one of about 20 vaccines under development to fight the disease that kills more one million people every year.
For decades, researchers have struggled, without much success, to find an effective vaccine against the parasite that causes malaria. The malaria parasite is said to be tenacious. Parasites change their skin much faster than virus and bacteria do. A report by researchers at the United States National Institutes of Health, which was published in the Lancet Infectious Disease in 2012, stated that 35 per cent of antimalarial medicines were either sub-standard or did not contain enough of the active principles to cure the disease.
But RTS,S is the first vaccine to provide protection against parasitic diseases. The clinical tests for the vaccine, which are currently in phase III, are being carried out on 16,000 children aged between five and 17 months. By the way, statistics have shown that 85 per cent of those who die from malaria are pre-school children of which African countries account for more than 90 per cent. Apart from the human costs attributable to malaria, sub-Saharan African countries lose as much of $12 billion in Gross Domestic Product (GDP), which is more than the entire GDP of Rwanda.
Blaise Genton, Project leader at the Swiss Tropical and Public Health Institute in Basel, and who is involved in the field tests of RTS,S, says, “When you are working in malaria, if you halve the problem, you feel you’ve gone a long way.” Although Genton admits that 50 per cent may not be enough, it signifies a lot of progress.
Bernard Pécoul who is Geneva-based executive director of the Drugs for Neglected Diseases initiative (DNDi), says “The important thing is that we have shown for the first time that a vaccine can work. This is a fundamental scientific breakthrough.”
The vaccine itself is not a panacea. It is part of the global efforts geared towards ridding the world of the deadly disease that is endemic in many developing countries, especially in Africa. The other approaches to fighting the disease is by eliminating the anopheles mosquitos, which are the carriers of the parasite when they ingest it after taking a blood meal from an infected person. Alternatively, the mosquitoes can be stopped from biting through use of treated mosquito nets. Another approach to fighting malaria is by effective treatment of infected persons to prevent the parasite from being pass on others.
RTS,S was developed by GlaxoSmithKline (GSK), with the support from the Bill and Melinda Gates Foundation. The Foundation has supported research carried out by Medicines for Malaria Venture foundation with millions of dollars. In terms of what the vaccine will cost, no price has been fixed but the company has assumed that it will be affordable so that it can be accessible to those who need it.
Blaise Genton also says, “it’s possible to reduce malaria if we are give ourselves the means to do so." He calls on governments of countries where malaria is prevalent to continue to regards malaria as a public health issue.
Related
-
West African leaders pledge to implement integrated approach to public health
By taking an integrated approach, communities can identify outbreaks in animals before they spread to humans.
-
Access Bank distributes mosquito nets to pregnant women
The campaign to reduce malaria burden among pregnant women reached 200,000 people online.
-
Malaria’s deadly comeback
Artemisinin resistance could move into India and onward to Sub-Saharan Africa and the rest of the world, putting millions ...